|
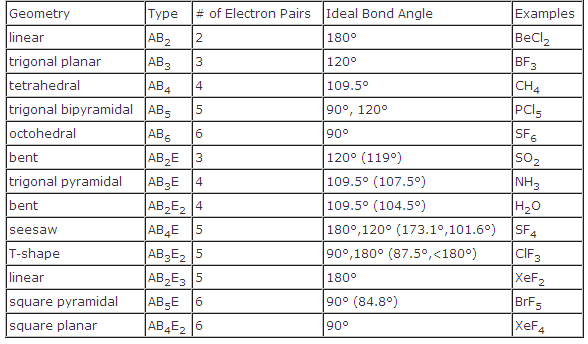
What is CLF3 molecular geometry?ĬlF3 has a T-shaped molecular geometry and trigonal bipyramidal electron geometry. It is the main precursor of sulfuric acid and is naturally present in tiny levels in the environment. When SO2 is dissolved in water, it produces a weak acid solution. It is a colorless, poisonous, inorganic gas with a strong odor similar to nitric acid. SO2 (Sulfur dioxide) is the result of a link between the atoms of sulfur and oxygen. They are found in the outermost electron shell of atoms and can be identified by drawing lewis’s structure. They are also called lone pairs of electrons. Unbonded pairs of electrons are unshared valence electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
